FDA OKs appliance that’s made in Indiana
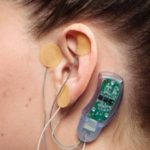 GREENWOOD — Since February, Greenwood City Court Judge Lewis Gregory has offered plan through the local probation department in which opiate addicts can choose to wear a NSS-2 Bridge, a hearing aid-sized device that helps fight the pain of withdrawal.
GREENWOOD — Since February, Greenwood City Court Judge Lewis Gregory has offered plan through the local probation department in which opiate addicts can choose to wear a NSS-2 Bridge, a hearing aid-sized device that helps fight the pain of withdrawal.
The other option is often jail.
“The attraction of the Bridge is that people can go through detox without confinement either in a jail or a treatment bed,” Gregory said. “With the number of folks we have in Indiana with an opioid problem, we really need that option.”
This week, the U.S. Food and Drug Administration approved the Indiana-produced Bridge as the first non-narcotic electronic device to help reduce withdrawal symptoms.
On Thursday, state Sen. Jim Merritt, R-Indianapolis, called it “the future of recovery.”
As a leader in presenting legislation to fight the opioid crisis, Merritt has touted the device’s promising aspects for months.
“This is something that’s going to be an incredible device and something I think will revolutionize the country in the way of addiction and the epidemic in our world,” Merritt said.
“People will detox, they will withdraw from drugs if there is a simpler process, and this is it.”
The NSS-2 Bridge was developed by Innovative Health Solutions, based in Versailles. It is manufactured by Key Electronics in Jeffersonville, where developers are hoping to produce 40,000 devices a year.
FDA Commissioner Scott Gottlieb said in a statement: “While we continue to pursue better medicines for the treatment of opioid use disorder, we also need to look to devices that can assist in this therapy.”
The NSS-2 Bridge is a small electrical nerve stimulator placed behind a patient’s ear. It contains a battery-powered chip that emits electrical pulses to stimulate branches of certain cranial nerves. It has been used in acupuncture and other pain relieving procedures.
But researchers found that the stimulations can offer relief from opioid withdrawal symptoms. Patients use the device for up to five days during the acute physical withdrawal phase, which often includes sweating, nervousness, insomnia and joint pain.
The device was tested on 73 patients, the FDA said. All had a reduction in symptoms within 30 minutes of using it. Overall, 64 of the patients moved to medication therapy after five days.
Similar results are true in the 13 cases it has been used in Gregory’s court. Of those, 10 people transitioned to Vivitrol, which blocks opioid receptors in the brain. People in three other cases did not transition but it was not due to the device, Gregory said.
Addicts going through the Greenwood court are encouraged to admit their problem to their probation officer, who then arranges for use of the Bridge. Gregory had hoped for two or three participants a week but because it is a voluntary program, it has worked out to two a month.
The device costs $499 and with installation runs about $1,000, Merritt said. Developers are hoping that the costs will eventually be covered by health insurance plans.
